If you like chemistry, than you'll be excited to hear that I'm starting a unit on it! I'll be doing a series called "Meet the Elements", and today is the first post. The star element: potassium.
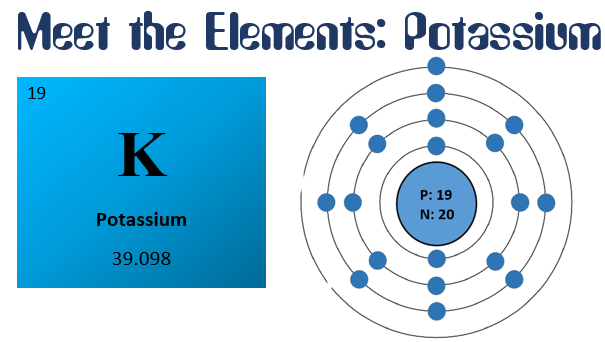
If you’re looking for potassium, you’ll find it in the fourth period and the first family of the periodic table, in the metals section. The box will be labeled with a 19 in the corner, which is the atomic number; a K in the center, which is potassium’s symbol; and a 39.098 at the bottom, which is the atomic mass.
If you were to, somehow, look at the atoms in potassium, you would find 19 protons and 20 neutrons in the nucleus. Circling around the nucleus, you would find 19 small electrons, located on different paths called energy shells- 2 on the first shell, 8 on the second shell, 8 on the third shell, and 1 on the last shell, called the valence electron.
The element potassium is a silver-whitish, soft metal that oxidizes when exposed to air. Its melting point is 63°C or 145°F, which is very low for a metal; you could hold it over the flame of a candle and it would melt. Potassium’s boiling point is 770°C or 1,420°F. Potassium is less dense than water, so it would float in water, but I wouldn’t recommend testing that; potassium is very active and reacts violently with water.
As a pure element, potassium has few uses. But its compounds, specifically potassium chloride, are used often in fertilizers. Other potassium compounds are used in products such as baking powders, soft drinks, fire extinguishers, explosives, fireworks, matches, dyes, rocket fuel, detergents, and liquid soaps.
If you were to, somehow, look at the atoms in potassium, you would find 19 protons and 20 neutrons in the nucleus. Circling around the nucleus, you would find 19 small electrons, located on different paths called energy shells- 2 on the first shell, 8 on the second shell, 8 on the third shell, and 1 on the last shell, called the valence electron.
The element potassium is a silver-whitish, soft metal that oxidizes when exposed to air. Its melting point is 63°C or 145°F, which is very low for a metal; you could hold it over the flame of a candle and it would melt. Potassium’s boiling point is 770°C or 1,420°F. Potassium is less dense than water, so it would float in water, but I wouldn’t recommend testing that; potassium is very active and reacts violently with water.
As a pure element, potassium has few uses. But its compounds, specifically potassium chloride, are used often in fertilizers. Other potassium compounds are used in products such as baking powders, soft drinks, fire extinguishers, explosives, fireworks, matches, dyes, rocket fuel, detergents, and liquid soaps.
 RSS Feed
RSS Feed
